Adapting HPV vaccine programmes in the COVID-19 pandemic
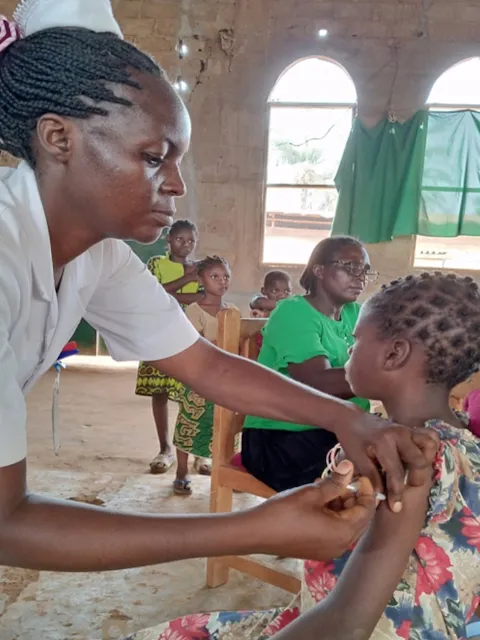
When the COVID-19 pandemic first hit and schools closed, girls in Uganda weren’t only missing out on in-person learning but also access to HPV vaccination. An inside look from Tara Newton at PATH on the occasion of World Immunization Week, 24-30 April.
Cervical cancer is a leading cause of cancer death among women in low- and middle-income countries (LMICs). More than 604,000 new cases and 341,000 deaths occur annually, with nearly 90% of deaths occurring in LMICs. Human papillomavirus (HPV) vaccines are highly effective and, since introduction, have significantly reduced vaccine-type HPV infections and precancerous cervical lesions.
Although HPV vaccine coverage is increasing, only 15% of girls worldwide are fully protected and 70% of the target population of girls 9-14 years of age live in countries without a national HPV vaccine programme. Uptake in LMICs has been limited due to barriers such as high programmatic costs, complex delivery strategies, supply shortages, vaccine hesitancy and, most recently, the COVID-19 pandemic.
Countries adapt to the pandemic – the case of Uganda
Countries are currently faced with an enormous burden of COVID-19 response and vaccine roll out in a dynamic environment. The pandemic has caused many disruptions to essential health services, including routine immunisation. Two countries where PATH provides technical assistance for HPV vaccine successfully adapted their programmes.
When Uganda went into lockdown, the government recognised that the HPV vaccine programme had to shift from school-based to community-focused. To pivot the programme, our PATH Uganda team advised and mentored health workers to intensify mobilisation and sensitisation for HPV vaccination, both at health facilities and other health service points. They worked closely with health workers and community resource people to identify and reach girls who missed either their first or second dose of HPV vaccine.
Vaccine hesitancy is also a concern (and now perpetuated by misinformation surrounding the COVID-19 vaccine) PATH recently partnered with the ministry of health to support a radio talk show for women with children who had received the HPV vaccine in 2007-8 to dispel rumours that the vaccine could cause fertility issues. The evidence shared during the programme by health officials and mothers helped strengthen the public’s trust in the vaccine.
In Mauritania, the national HPV vaccine launch planned for 2020 was delayed due to COVID-19. Despite the ongoing pandemic and stretched country resources, our team partnered with the Expanded Program on Immunization (EPI), Gavi, World Health Organization (WHO) and UNICEF, for a successful launch across the country to reach all 9–14-year-old girls in both schools and health facilities in March 2021. Our team helped with the introduction planning, training of health workers and trainers, data collection tools and microplanning. Currently, we are continuing to supervise the vaccination campaign.
Overcoming barriers to HPV vaccine access
In November 2020, the WHO launched the global strategy to accelerate the elimination of cervical cancer as a public health problem. In addition to strong programmes for screening, treatment and palliative care, one target of the strategy is for all countries to fully vaccinate 90% of girls aged 9-15 against HPV by 2030. Given the barriers countries are facing, how are we going to get there?
Last year, the WHO issued a provisional recommendation during the HPV vaccine supply constraint to extend the interval between doses up to three to five years, giving countries more flexibility for administering the second dose. Additionally, accumulating evidence suggests that a single dose of HPV vaccine may elicit an immune response that can protect against HPV infection. Several researchers in the Single-Dose HPV Vaccine Evaluation Consortium managed by PATH are generating, compiling and evaluating the evidence of a single-dose HPV vaccination schedule to inform global policy discussions and implications for national programmes. This evidence is particularly relevant as HPV vaccine programmes face increased challenges with vaccine accessibility and delivery in the current pandemic and supply shortage.
Access to HPV vaccines could also increase with more affordable vaccine options produced by developing country manufacturers. With several in the pipeline and one under WHO prequalification review, countries will soon have more options for their programs. Finally, PATH and several other organisations are conducting analyses on the cost-effectiveness of HPV vaccines and the cost to deliver HPV vaccines across several countries. Armed with this new data, countries will be able to make informed decisions on the value and programmatic implications of introducing or expanding an HPV vaccination programme.
Last update
Friday 23 April 2021Share this page